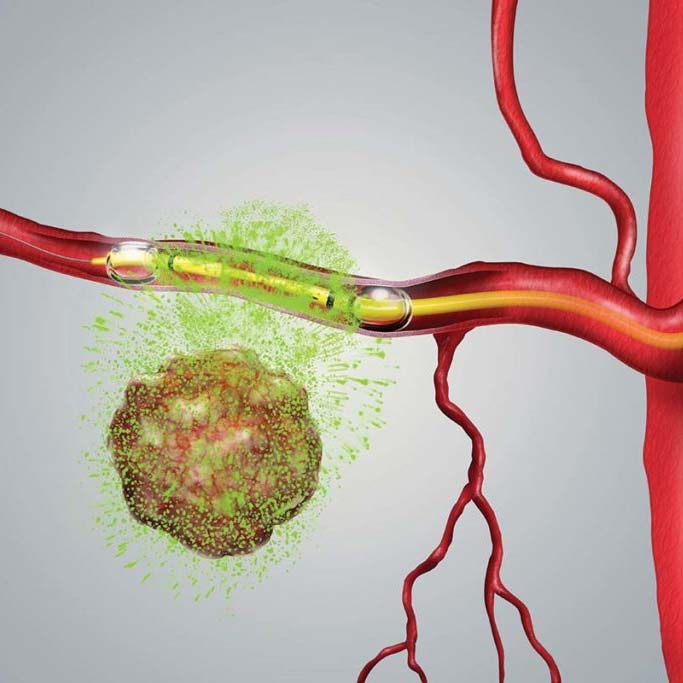
510(k) Device May Improve Targeted Delivery of Chemotherapy
Los Altos, CA, September 7, 2021 – RenovoRx, Inc. (Nasdaq: RNXT), a biopharmaceutical company and innovator in targeted cancer therapy, today announced it has received a new 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its proprietary RenovoCath Delivery System. The RenovoCath Delivery System is the device component of the Company’s initial product, RenovoGem™. This drug/device combination used in RenovoRx’s Trans-Arterial Micro-Perfusion (RenovoTAMP®) therapy platform is a dual-balloon infusion catheter delivering chemotherapy directly to tumors via arteries. This new design provides more targeted delivery of therapy which the Company believes can translate into more effective treatment with fewer side effects. RenovoRx received its initial 510(k) for the RenovoCath delivery system in 2014.
“Our novel RenovoTAMP therapy platform isolates the region of disease enabling delivery of the optimal dose of targeted chemotherapy to the tumor location,” said Shaun Bagai, Chief Executive Officer at RenovoRx. “We believe this new design cleared through the FDA 510(k) process will further improve RenovoTAMP’s targeted delivery. Our focus is maximizing patient survival while minimizing side effects for cancer patients.”
RenovoTAMP is the Company’s novel therapy platform designed to deliver well-established chemotherapeutic agents for localized treatment of solid cancer tumors. It is under clinical investigation for increasing survival and improving quality of life. This platform technology may enable physicians to isolate the anatomy and micro-perfuse targeted tissue with small molecule chemotherapy.
As of August 15, 2021, the Phase 3 TIGeR-PaC clinical trial — a randomized study utilizing the RenovoTAMP platform to evaluate RenovoGem — reached 44 percent patient enrollment for the treatment of pancreatic cancer. TIGeR-PaC is currently enrolling locally advanced, pancreatic cancer patients. In clinical studies conducted to date, chemotherapy delivered with the RenovoTAMP platform was associated with more than half the patients living more than two years. Pancreatic cancer patients treated standard-of-care typically live12 to 15 months after diagnosis.
The TIGeR-PaC study, which currently has approximately 30 active clinical sites, is expected to involve approximately 200 participants in the US and Europe. To learn more, visit https://renovorx.com/clinical-trial/.
About RenovoRx, Inc.
RenovoRx, a biopharmaceutical company headquartered in Silicon Valley, California, has developed a therapy platform targeting difficult-to-treat tumors. Targeted therapy via the proprietary RenovoRx Trans-Arterial Micro-Perfusion (RenovoTAMP) platform safely and without transmission to non-targeted areas, is the primary focus.
RenovoRx’s patent portfolio includes seven U.S. patents for its technology. The Company also has secured two separate Orphan Drug Designations from the FDA for intra-arterial gemcitabine including pancreatic cancer and bile duct cancer. The RenovoTAMP therapy is being studied in the Phase III TIGeR-PaC trial for the treatment of locally advanced pancreatic cancer.
RenovoRx won the Drug Delivery Technology category of the Fierce Innovation Awards – Life Sciences Edition 2020 for its RenovoTAMP technology.
Learn more by visiting the RenovoRx website or following us on Facebook, LinkedIn and Twitter.
Forward-Looking Statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. These statements may be identified using words such as “anticipate,” “believe,” “forecast,” “estimated” and “intend” or other similar terms or expressions that concern RenovoRx’s expectations, strategy, plans or intentions. These forward-looking statements are based on RenovoRx’s current expectations and actual results could differ materially. There are several factors that could cause actual events to differ materially from those indicated by such forward-looking statements. These factors include, but are not limited to, clinical trials involve a lengthy and expensive process with an uncertain outcome, and results of earlier studies and trials may not be predictive of future trial results; our clinical trials may be suspended or discontinued due to unexpected side effects or other safety risks that could preclude approval of our product candidate; risks related to business interruptions, including the outbreak of COVID-19 coronavirus, which could seriously harm our financial condition and increase our costs and expenses; uncertainties of government or third party payer reimbursement; dependence on key personnel; limited experience in marketing and sales; substantial competition; uncertainties of patent protection and litigation; dependence upon third parties; and risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations. There are no guarantees that any of our technology or products will be utilized or prove to be commercially successful. Investors should read the risk factors set forth in our registration statement on Form S-1 and our periodic reports filed with the Securities and Exchange Commission. While the list of factors presented here is considered representative, no such list should be considered to be a complete statement of all potential risks and uncertainties. Unlisted factors may present significant additional obstacles to the realization of forward-looking statements. Forward-looking statements included herein are made as of the date hereof, and RenovoRx does not undertake any obligation to update publicly such statements to reflect subsequent events or circumstances.
Contact: Chris Lehman, (650) 284-4433