RenovoRx Phase 3 TIGeR-PaC Clinical Trial Preliminary Pharmacokinetic Data Featured
Miami, FL and Los Altos, CA, November 9, 2021 – RenovoRx, Inc. (Nasdaq: RNXT), a biopharmaceutical company and innovator in targeted cancer therapy, today announced its novel therapy platform, RenovoTAMP™ (RenovoRx Trans-Arterial Micro-Perfusion), will be highlighted during a presentation by expert panelist, Dr. Ripal Gandhi, at the Miami Cancer Institute’s New Advances in the Management of Pancreatic Cancer CME course this evening.
Dr. Gandhi, a Principal Investigator in RenovoRx’s ongoing Phase 3 TIGeR-PaC study, and Professor of Interventional Radiology at the Miami Cancer Institute and Miami Cardiac and Vascular Institute, Florida International University Herbert Wertheim College of Medicine, will
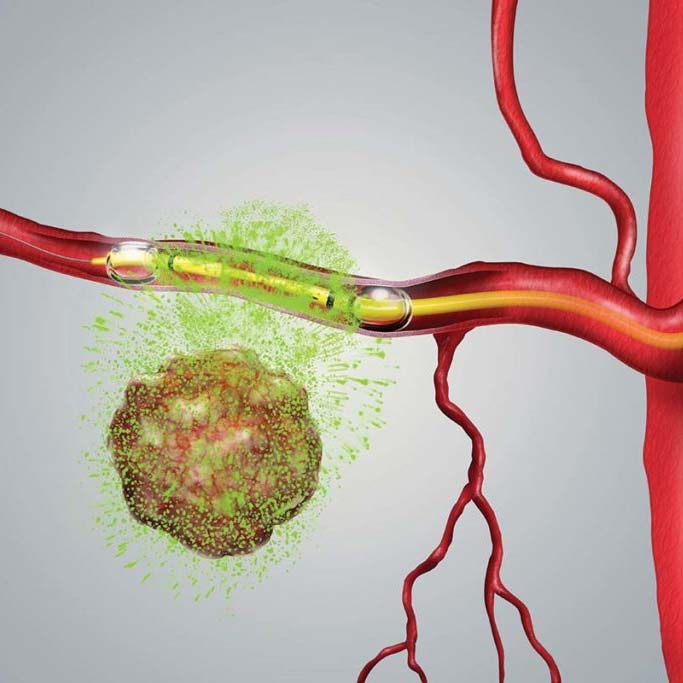
describe the results from initial clinical studies of RenovoTAMP used in combination with radiation therapy and its benefits when managing patients with stage 3 locally advanced pancreatic cancer (LAPC). He will also review the key stages of the TIGeR-PaC protocol which uses RenovoTAMP for the intra-arterial delivery of gemcitabine, an approved chemotherapeutic agent, to treat unresectable LAPC. To highlight a critical difference between intravenous (IV) and intra-arterial (IA) chemotherapy, Dr. Gandhi will present pharmacokinetic data, or data describing the drug absorption/distribution/metabolism/excretion of gemcitabine from five patients in the Phase 3 TIGeR-PaC study that received intra-arterial gemcitabine. The data demonstrate an approximately two-thirds reduction in systemic gemcitabine when compared to systemic levels in patients traditionally receiving IV infusion of gemcitabine.
“This innovative therapy platform is enabling the targeted treatment of inoperable LAPC with decreased side effects typical of systemic chemotherapy, while shifting the focus to what is most important to our patients: improving quality of life and allowing them to spend more time with their family and loved ones,” said Dr. Gandhi. “RenovoTAMP, when used in combination with radiation therapy, is designed to reduce arterial microvasculature, thereby minimizing leakage during drug delivery and enhancing drug delivery directly to the tumor.”
A copy and recording of Dr. Gandhi’s presentation, titled “Potential Future Innovative Strategy for Pancreatic Cancer: Intra-Arterial Chemotherapy,” will be available on RenovoRx’s website at https://renovorx.com.
About the Phase 3 TIGeR-PaC Clinical Trial
The TIGeR-PaC clinical trial is a randomized multi-center study using the RenovoTAMP™ platform to evaluate RenovoRx’s first product candidate, RenovoGem™ to treat unresectable LAPC through the intra-arterial delivery of gemcitabine, an approved chemotherapeutic agent. TIGeR-PaC is currently enrolling locally advanced, unresectable pancreatic cancer patients. To learn more about the study and the participating clinical trial sites, visit https://renovorx.com/clinical-trial/.
About RenovoRx, Inc.
RenovoRx is a clinical-stage biopharmaceutical company focused on fighting cancer through the localized treatment of difficult to treat tumors via its proprietary RenovoRx Trans-Arterial Micro-Perfusion (RenovoTAMPTM) therapy platform. RenovoTAMP delivers approved small molecule chemotherapeutic agents locally to the solid tumors. RenovoRx’s lead product candidate, RenovoGemTM, uses intra-arterial delivery of gemcitabine, an approved chemotherapeutic agent, to treat unresectable locally advanced pancreatic cancer (LAPC) and is currently being studied in the Phase 3 TIGeR-PaC trial for the treatment of LAPC.
RenovoRx’s patent portfolio includes seven U.S. patents for its technology. RenovoRx has been granted Orphan Drug Designation for intra-arterial delivery of gemcitabine for the treatment of both pancreatic cancer and bile duct cancer.
RenovoRx won the Drug Delivery Technology category of the Fierce Innovation Awards – Life Sciences Edition 2020 for its RenovoTAMP technology.
Learn more by visiting the RenovoRx website or following us on Facebook, LinkedIn and Twitter.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, and Section 21E of the Securities Exchange Act of 1934, including but not limited to statements regarding our Phase 1 (RR1) and Observational Registry (RR2) studies, statements regarding the potential of RenovoTAMPTM, RenovoCath® or RenovoGemTM or regarding our ongoing TIGeR-PaC Phase 3 clinical trial in LAPC, and statements regarding the potential for our product candidates to treat or provide clinically meaningful outcomes for certain medical conditions or diseases. Statements that are not purely historical are forward-looking statements. The forward-looking statements contained herein are based upon our current expectations and beliefs regarding future events, many of which, by their nature, are inherently uncertain, outside of our control and involve assumptions that may never materialize or may prove to be incorrect. These may include estimates, projections and statements relating to our research and development plans, clinical trials, therapy platform, business plans, objectives and expected operating results, which are based on current expectations and assumptions that are subject to known and unknown risks and uncertainties that may cause actual results to differ materially from those expressed or implied by these forward-looking statements. These statements may be identified using words such as “may,” “expects,” “plans,” “aims,” “anticipates,” “believes,” “forecasts,” “estimates,” “intends,” and “potential,” or the negative of these terms or other comparable terminology regarding RenovoRx’s expectations strategy, plans or intentions, although not all forward-looking statements contain these words. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, that could cause actual events to differ materially from those projected or indicated by such statements, including, among other things: the timing of the initiation, progress and potential results of our preclinical studies, clinical trials and our research programs; our ability to use and expand our therapy platform to build a pipeline of product candidates; our ability to advance product candidates into, and successfully complete, clinical trials; the timing or likelihood of regulatory filings and approvals; our estimates of the number of patients who suffer from the diseases we are targeting and the number of patients that may enroll in our clinical trials; the commercialization potential of our product candidates, if approved; our ability and the potential to successfully manufacture and supply our product candidates for clinical trials and for commercial use, if approved; future strategic arrangements and/or collaborations and the potential benefits of such arrangements; our estimates regarding expenses, future revenue, capital requirements and needs for additional financing and our ability to obtain additional capital; the sufficiency of our existing cash and cash equivalents to fund our future operating expenses and capital expenditure requirements; our ability to retain the continued service of our key personnel and to identify, hire and retain additional qualified personnel; the implementation of our strategic plans for our business and product candidates; the scope of protection we are able to establish and maintain for intellectual property rights, including our therapy platform, product candidates and research programs; our ability to contract with third-party suppliers and manufacturers and their ability to perform adequately; the pricing, coverage and reimbursement of our product candidates, if approved; developments relating to our competitors and our industry, including competing product candidates and therapies; negative impacts of the COVID-19 pandemic on our operations; and other risks.
Information regarding the foregoing and additional risks may be found in the section entitled “Risk Factors” in documents that RenovoRx files from time to time with the Securities and Exchange Commission.
Forward-looking statements included herein are made as of the date hereof, and RenovoRx does not undertake any obligation to update publicly such forward-looking statements to reflect subsequent events or circumstances, except as required by law.
Contact: Chris Lehman, (650) 284-4433