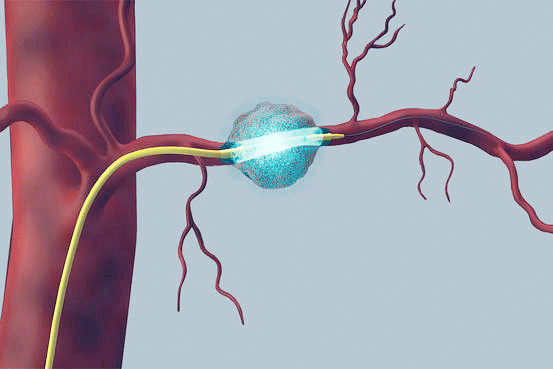
Aim: A dose-escalation study of Gemcitabine for treatment of unresectable pancreatic cancer, using a targeted intra-arterial delivery catheter (RenovoCath™).
Methods: 20 patients at two centers were enrolled with a fourstage dose escalation of gemcitabine up to 1000 mg/m2. Enzyme markers, blood count, and constitutional endpoints were monitored to assess for dose-limiting toxicities. Feasibility and safety of repeated intra-arterial treatment sessions were assessed. Secondary endpoints included the effect on tumor size by imaging, tumor markers, and conversion to resectability.
Results: Data for 20 patients enrolled is presented. Efficacy analysis was limited to the 15 patients who received more than two treatments.
SUMMARY: Intra-arterial delivery of gemcitabine using localized delivery catheter, RenovoCath, is safe provided:
- Patients with prior biliary stent/drain receive peri-op antibiotics
- IR training/support is necessary to avoid guide mediated vascular complications
- Early efficacy results of survival is encouraging, especially in patients with prior radiation where there may exist a synergistic effect with localized trans-arterial delivery of gemcitabine